The Incidence and Risk Factors for Psoriatic Arthritis in Patients With Psoriasis: A Prospective Cohort Study
Abstract
Objective
To estimate the incidence of psoriatic arthritis (PsA) in patients with psoriasis, and to identify risk factors for its development.
Methods
The study was designed as a prospective cohort study involving psoriasis patients who did not have a diagnosis of arthritis at the time of study enrollment. Information was collected about lifestyle habits, comorbidities, psoriasis activity, and medications. Patients who developed inflammatory arthritis or spondylitis were classified as having PsA if they fulfilled the criteria of the Classification of Psoriatic Arthritis Study group. The annual incidence of PsA was estimated using an event per person‐years analysis. Cox proportional hazards models, involving fixed and time‐dependent explanatory variables, were fitted to obtain estimates of the relative risk (RR) of the onset of PsA, determined in multivariate models stratified by sex and controlled for age at onset of psoriasis.
Results
The data obtained from the 464 patients who were followed up for 8 years were analyzed. A total of 51 patients developed PsA during the 8 years since enrollment. The annual incidence rate of PsA was 2.7 cases (95% confidence interval 2.1–3.6) per 100 psoriasis patients. The following baseline variables were associated with the development of PsA in multivariate analysis: severe psoriasis (RR 5.4, P = 0.006), low level of education (university/college versus high school incomplete RR 0.22, P = 0.005; high school graduate versus high school incomplete RR 0.30, P = 0.049), and use of retinoid medications (RR 3.4, P = 0.02). In multivariate models with time‐dependent variables, psoriatic nail pitting (RR 2.5, P = 0.002) and uveitis (RR 31.5, P = 0.0002) were associated with the development of PsA.
Conclusion
The incidence of PsA in patients with psoriasis is higher than previously reported. A severe psoriasis phenotype, presence of nail pitting, low level of education, and uveitis are predictive of the development of PsA in patients with psoriasis.
Psoriasis is a chronic immune‐mediated skin disease affecting ∼2% of the general population 1. Psoriatic arthritis (PsA) is an inflammatory arthritis that affects ∼20–30% of patients with psoriasis attending dermatology clinics 2. Despite major advances in the treatment of PsA over the past decade, many patients with PsA still experience decreased quality of life, disability, comorbidities, and extraarticular manifestations related to their disease. Early diagnosis and treatment are critical in the prevention of adverse disease‐related outcomes.
The relationship between psoriasis and PsA is not well understood. Epidemiologic studies have found that the majority of PsA patients first develop psoriasis and only later develop PsA 3. Therefore, patients with psoriasis may serve as a preferred target population for investigating the epidemiology of PsA and for identifying markers of high risk for the disease. Such information may contribute to the understanding of the pathophysiology of PsA and can also be used to develop clinical prediction algorithms that would help dermatologists identify psoriasis patients who are at high risk for developing PsA.
There is limited information about the incidence of PsA and risk factors for the disease in patients with psoriasis. The estimated cumulative incidence of PsA in psoriasis patients ranges widely, from 5.1% at 20 years 4 to 20.5% at 30 years 5 following the onset of psoriasis. In a prospective cohort study from our group, involving patients with psoriasis who did not have arthritis at baseline, the annual incidence rate of PsA was 1.87% 6. This estimate was much higher than that in previous reports and may reflect the systematic assessment of the cohort participants, which likely allowed the detection of milder cases. However, that study was based on a relatively short follow‐up period and small number of incident cases.
Screening for symptoms and signs of PsA in patients with psoriasis is recommended in dermatology guidelines 7, 8. Therefore, the identification of markers for an increased risk of developing PsA in this patient population is highly desirable. The presence of psoriatic nail lesions and severe psoriasis, as indicated by a more extensive area of the body surface being affected, were found to be associated with a higher risk of having PsA 4, 9-11. Involvement of certain body sites, particularly the scalp and intergluteal area, was associated with an increased risk of developing PsA in a retrospective cohort study 4. Obesity was predictive of the development of PsA in 2 large population‐based studies 12, 13. Other suggested environmental risk factors that have been associated with the development of PsA include trauma and physically demanding occupations, bacterial infections, smoking, and a family history of PsA 14-17. However, the evidence to support a link between PsA and these environmental risk factors is weaker and the results are often conflicting. Furthermore, previous studies concerning risk factors for PsA were limited by their retrospective nature, cross‐sectional study design, small sample size, a failure to use classification criteria for PsA, and the lack of a comprehensive assessment of psoriasis patients for detection of inflammatory arthritis, which may have led to misclassification of the 2 groups.
A prospective cohort study of patients who are at high risk for developing a condition can serve as an ideal setting for the investigation of preclinical phases of a disease. In this study, we report the results of 8 years of follow‐up of a prospective cohort of patients with psoriasis who were free of PsA at baseline. The objectives of this analysis were 1) to estimate the annual incidence of the development of PsA in patients with psoriasis, and 2) to identify markers of high risk for PsA in psoriasis patients.
PATIENTS AND METHODS
Setting
The Toronto Psoriasis Cohort was established in 2006 and forms the basis of a long‐term prospective study aimed at assessing risk factors for the development of PsA in patients with psoriasis. All potential study subjects have a diagnosis of psoriasis confirmed by a dermatologist. The sources of recruitment are varied and include patients with a range of psoriasis types (primarily, chronic plaque psoriasis) and varying degrees of disease severity. Patients are mainly recruited from dermatology clinics and phototherapy centers in the Greater Toronto Area and also from family practice clinics and through advertisement in flyers posted in several hospitals and local media. The cohort includes different ethnic groups, although most of the patients (77.3%) are white. The inclusion criteria require a diagnosis of psoriasis that is confirmed by a dermatologist. The exclusion criteria are the presence of inflammatory arthritis or spondylitis in the past or at the time of the assessment.
All subjects are evaluated by rheumatologists with expertise in assessment of PsA patients, in order to exclude a diagnosis of inflammatory arthritis or spondylitis before enrollment. Each subject undergoes a comprehensive musculoskeletal examination that includes joint assessment for tenderness, swelling, and deformities, evaluation for the presence of enthesitis, tendinitis, and dactylitis, and assessment for restriction of movement in the spine. If there are definite clinical findings of inflammatory arthritis, enthesitis, or spondylitis, the patient is excluded from the study. In cases of doubt, imaging studies, including radiographs, ultrasound, or magnetic resonance imaging (MRI), are performed, as indicated, to investigate the nature of the abnormality. Patients thus diagnosed with psoriasis who have no current evidence or history of PsA are eligible for the study. If a noninflammatory condition, such as osteoarthritis, is diagnosed, the subject is included in the study. This process ensures that none of the participants have clinical inflammatory arthritis at the time of study enrollment.
Case definition
All study participants were reassessed annually regardless of whether they had developed musculoskeletal symptoms. The process of incident case ascertainment for patients who developed musculoskeletal symptoms was similar to that described above for the baseline visit. The diagnosis of PsA was determined by at least 2 rheumatologists from the PsA research team after reviewing the clinical, laboratory, and imaging data. Participants were classified as having confirmed PsA if they fulfilled the criteria of the Classification of Psoriatic Arthritis Study group.
Patients who failed to return for the yearly assessment were requested by telephone or by mail to complete the Toronto Psoriatic Arthritis Screen II (ToPAS‐II) questionnaire, a screening questionnaire designed to detect PsA among patients with psoriasis as well as the general population 18. The ToPAS‐II was validated in different populations of patients with skin conditions other than psoriasis, patients with PsA, and healthy subjects 18. Subjects scoring ≥8 points on the ToPAS‐II screen were classified as having suspected PsA.
Data collection
Study participants were assessed according to a standard protocol at each visit. The subjects provided information about their demographics, lifestyle habits, family history of psoriasis and rheumatic diseases, comorbidities, and extraarticular manifestations of spondyloarthritis (SpA), injuries, infections, musculoskeletal symptoms, and the use of medications. The general physical examination included measurement of height and weight, assessment of psoriasis severity using the Psoriasis Area and Severity Index (PASI) 19, and assessment of the presence and type of psoriatic nail lesions (pitting or onycholysis). A comprehensive musculoskeletal assessment of 66 joints for swelling and 68 joints for tenderness was conducted. The presence and the number of digits with dactylitis were recorded. The presence of enthesitis in 16 sites was assessed using the Spondyloarthritis Research Consortium of Canada Enthesitis Index 20. To estimate the effect of psoriasis and/or PsA on function and quality of life, the following patient‐reported outcomes were assessed: Health Assessment Questionnaire 21, the Medical Outcomes Study Short Form 36 health survey 22, and the Dermatology Life Quality Index 23. Assessment of radiographic joint damage was performed at the time of diagnosis in the majority of patients who were confirmed to have an incident case of PsA. Radiographic damage in the hands and feet was assessed according to the modified Steinbrocker score 24. Axial radiographs were scored according to the modified Stoke Ankylosing Spondylitis Spine Score 25. MRI of the spine was performed in patients with inflammatory back pain who had negative or inconclusive findings on radiographs.
Statistical analysis
Descriptive statistics were computed. Continuous variables were expressed as the mean ± SD and categorical variables as proportions. The number of person‐years at risk was calculated as the time between the diagnosis of psoriasis and last contact date (follow‐up visit, date of ToPAS‐II questionnaire completion, or death) or the date of PsA diagnosis, whichever came first. This duration was used to estimate the cumulative incidence of PsA among patients with psoriasis. Patients who did not have any follow‐up visits or ToPAS‐II questionnaire assessments after their initial screening visit were excluded, since they did not contribute any time at risk.
Two parametric models were used to evaluate whether the risk of developing PsA, as modeled by the hazard function, changes over time. A time homogeneous exponential model (with a constant hazard function) was used to estimate the rate of PsA among psoriasis patients. This model was compared to a Weibull model that accommodates a trend in the hazard. Data from a likelihood ratio test of the 2 models were used to indicate lack of evidence of a time‐dependent risk. A nonparametric estimate of the cumulative incidence function conveyed the risk of PsA over time.
For the analysis of predictors of PsA, only subjects with complete information about all of the assessed predictors were included. For the primary analyses, the outcome included incident cases of PsA (confirmed and suspected). A last observation carried forward strategy was used to handle missing covariates during the follow‐up period. Cox proportional hazards models were used to assess predictors of the development of PsA. A total of 22 predictors were assessed, selected on the basis of knowledge from prior literature about the epidemiology and pathogenesis of PsA. Since this was considered an exploratory analysis, we did not account for multiple testing. Explanatory variables included a family history of psoriasis in a first‐ or second‐degree relative, family history of PsA or ankylosing spondylitis (AS) in a first‐ or second‐degree relative, uveitis, depression, hypothyroidism or hyperthyroidism, diabetes, presence of diarrhea, being in menopause (women only), alcohol consumption (daily, social, none), smoking status (current, past, never), injury (within 1 year), infection (within 1 year), infection that required antibiotic therapy (within 1 year), body mass index, nail pitting, onycholysis, psoriasis severity (PASI score >20 = severe, PASI score 10–20 = moderate, PASI score <10 = mild), level of education (university/college education, high school graduate, high school incomplete), use of methotrexate, use of tumor necrosis factor blockers, and use of retinoids.
As a screening phase, each of the above‐mentioned variables was included in a separate regression model that was controlled for age at onset of psoriasis and stratified by sex, with use of the duration of psoriasis as the left‐truncation time. Each explanatory variable was assessed twice: as a time‐varying covariate, and as a fixed variable at baseline. All variables that showed a significant association with PsA, based on a 10% level of significance, were included in a single multivariate Cox regression model that was controlled for age at onset of psoriasis and stratified by sex, with duration of psoriasis as the left‐truncation time. Backward elimination was used to eliminate nonsignificant covariates (based on a 5% level of significance) from the multivariate regression model. In addition, sensitivity analyses that considered only confirmed cases of PsA as an outcome were carried out. Suspected cases of PsA were censored at the last clinic visit.
RESULTS
Follow‐up summary
A total of 631 patients with psoriasis were screened. Of those, 60 patients were excluded due to the presence of PsA and 13 patients were excluded due to the presence of other rheumatic conditions at baseline. The characteristics of the excluded patients with PsA at baseline were similar to the baseline characteristics of the study population, except that the group of excluded patients had a higher proportion of men and a higher proportion of subjects with psoriatic nail lesions (details available upon request from the corresponding author). A total of 558 patients who were followed up from January 1, 2006 to September 5, 2014 were enrolled in the Toronto Psoriasis Cohort. Of those, 94 subjects were excluded from the analysis since they only had a single assessment and did not contribute any data in the follow‐up period (81 were lost to follow‐up and 13 declined to return for a follow‐up visit or to complete the ToPAS‐II questionnaire). The patients who were excluded tended to be younger than the remaining study population, but were similar to the study population with respect to other characteristics.
Of the 464 patients who were included in the final analysis, 51 patients developed PsA, as confirmed by a rheumatologist (confirmed cases), and an additional 9 patients were considered to have suspected PsA, based on a high score on the ToPAS‐II questionnaire. The subjects in the latter group, however, declined to return for follow‐up; therefore, the diagnosis of PsA could not be confirmed. The remaining 404 patients were free of PsA at their last assessment. This cohort had a total of 1,880.9 person‐years of follow‐up, with a mean ± SD 4.1 ± 2.1 years of follow‐up per person. The distribution of the study population is summarized in Figure 1.
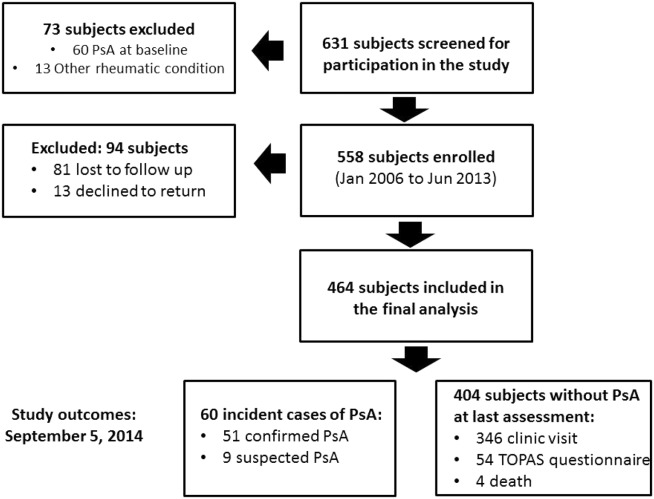
Follow‐up summary of the study population. PsA = psoriatic arthritis; TOPAS = Toronto Psoriatic Arthritis Screen II.
Characteristics of the study population
Most of the participants were recruited from phototherapy centers (66.4%). The rest were recruited from local advertisement (19.2%), from dermatology clinics (11.2%), and from a general rheumatology clinic (3.2%). The latter group included patients who were referred to a rheumatologist for assessment of musculoskeletal abnormalities but were found to have a noninflammatory musculoskeletal condition. The characteristics of the study population are summarized in Table 1. Their mean ± SD age at baseline was 47.2 ± 13.3 years, and 56.2% of the study subjects were men. The mean ± SD duration of psoriasis at baseline was 16.4 ± 14.4 years, and 74.4% had early‐onset psoriasis (prior to the age of 40 years). The majority of the participants were not taking any systemic medications for their psoriasis.
| All patients (n = 464) | Patients with incident PsA (n = 60)b | |
|---|---|---|
| Age, mean ± SD years | 47.2 ± 13.3 | 46.7 ± 12.4 |
| Sex, male | 261 (56.2) | 33 (55) |
| Psoriasis duration, mean ± SD years | 16.4 ± 14.4 | 17 ± 15.2 |
| Psoriasis onset prior to age 40 years | 345 (74.4) | 45 (75) |
| Family history of psoriasis | 189 (40.7) | 28 (46.7) |
| Family history of PsA or AS | 15 (3.2) | 3 (5) |
| Smoking status | ||
| Current smoker | 104 (22.4) | 14 (23.3) |
| Past smoker | 155 (33.4) | 22 (36.7) |
| Never | 205 (44.2) | 24 (40) |
| Alcohol consumption | ||
| Daily | 59 (12.9) | 7 (12.1) |
| Social | 259 (56.8) | 35 (60.3) |
| None | 138 (30.3) | 16 (27.6) |
| Level of education | ||
| University/college | 369 (79.5) | 43 (71.7) |
| High school graduate | 69 (14.9) | 12 (20) |
| High school incomplete | 26 (5.6) | 5 (8.3) |
| PASI score | ||
| <10 (mild) | 403 (86.9) | 47 (78.3) |
| 10–20 (moderate) | 50 (10.8) | 9 (15) |
| >20 (severe) | 11 (2.4) | 4 (6.7) |
| Nail pitting | 166 (36) | 33 (55.9) |
| Nail onycholysis | 151 (32.8) | 26 (44.1) |
| Use of retinoids ever | 44 (9.5) | 8 (13.3) |
| Use of methotrexate ever | 44 (9.5) | 9 (15) |
| Use of TNF blockers ever | 31 (6.7) | 6 (10) |
| BMI | ||
| Obese | 127 (28.5) | 26 (43.3) |
| Overweight | 174 (39.1) | 18 (30) |
| Normal | 144 (32.4) | 16 (26.7) |
| Uveitis ever | 5 (1) | 3 (5) |
| Inflammatory bowel disease ever | 4 (0.9) | 1 (1.7) |
| Diabetes | 34 (7.3) | 4 (6.7) |
| Thyroid disease | 38 (8.2) | 8 (13.3) |
| Depression | 35 (7.5) | 5 (8.3) |
- a The reported variables are from the baseline visit, except when “ever” is indicated, which means the variable was present at any time during the study period. Except where indicated otherwise, values are the number (%) of patients. AS = ankylosing spondylitis; PASI = Psoriasis Activity and Severity Index; TNF = tumor necrosis factor; BMI = body mass index.
- b Includes 51 confirmed cases and 9 suspected cases of psoriatic arthritis (PsA).
Characteristics of PsA patients at diagnosis
The clinical features of the 51 psoriasis patients whose diagnosis of PsA was confirmed are presented in Table 2. The mean ± SD age at the time of diagnosis was 49.9 ± 12.8 years. The most frequent pattern of musculoskeletal inflammation at presentation was peripheral arthritis (64.7%) followed by axial arthritis (17.6%), peripheral and axial arthritis (15.7%), and enthesitis (2%). The majority of the patients with peripheral involvement (32 [76.2%] of 42) had oligoarthritis (4 or fewer actively inflamed joints) at the time of diagnosis, and 41.4% of the patients had signs of sacroiliitis or spondylitis on imaging (radiographs or MRI). In total, 25% of patients were found to have radiographic sacroiliitis (bilateral grade 2 or unilateral grade 3 sacroiliitis), while in 4 (44.4%) of the 9 patients who presented with inflammatory back pain without peripheral arthritis, the diagnosis was confirmed by MRI of the spine (nonradiographic axial SpA). Radiographs of the hands and feet were performed in 39 patients. Of these patients, 23% were found to have at least one periarticular joint erosion and 10.2% had periarticular new bone formation.
| Age at PsA diagnosis, mean ± SD years | 49.9 ± 12.8 |
| Pattern of joint involvement | |
| Peripheral | 33 (64.7) |
| Axial | 9 (17.6) |
| Peripheral and axial | 8 (15.7) |
| Enthesitis alone | 1 (2) |
| Pattern of peripheral arthritis | |
| Polyarthritis | 10 (23.8) |
| Oligoarthritis | 32 (76.2) |
| Tender joint count, mean ± SD | 2.8 ± 4.5 |
| Swollen joint count, mean ± SD | 1.5 ± 1.8 |
| Enthesitis | 10 (19.6) |
| Dactylitis | 3 (5.9) |
| Psoriatic nail lesions | 33 (64.7) |
| Peripheral joint erosions | 9 (23) |
| Sacroiliitis or spondylitis | |
| By radiography | 9 (21.9) |
| By radiography or MRI | 17 (41.4) |
| Periarticular new bone formation | 4 (10.2) |
| HAQ score, mean ± SD (scale 0–3) | 0.34 ± 0.43 |
| Pain score (VAS scale 0–10), mean ± SD | 3.28 ± 2.50 |
| Patient global assessment of disease activity (VAS scale 0–10), mean ± SD | 4.02 ± 2.44 |
| DLQI score, mean ± SD (scale 0–30) | 6.77 ± 6.14 |
- a Except where indicated otherwise, values are the number (%) of patients. MRI = magnetic resonance imaging; HAQ = Health Assessment Questionnaire; VAS = visual analog scale; DLQI = Dermatology Life Quality Index.
Incidence of PsA
The annual incidence rate of PsA was 2.7 cases (95% confidence interval [95% CI] 2.1–3.6) per 100 psoriasis patients, when only confirmed cases were included. The incidence rate increased to 3.2 cases (95% CI 2.5–4.1) when both confirmed and suspected cases were included. The cumulative incidence function for PsA onset, with death as a competing event, and the estimated cumulative probability of developing PsA based on the exponential model are shown in Figure 2. Tests for trend did not suggest a departure from the constant hazard, and therefore there is insufficient evidence to claim that the risk of developing PsA in patients with psoriasis changes over time.
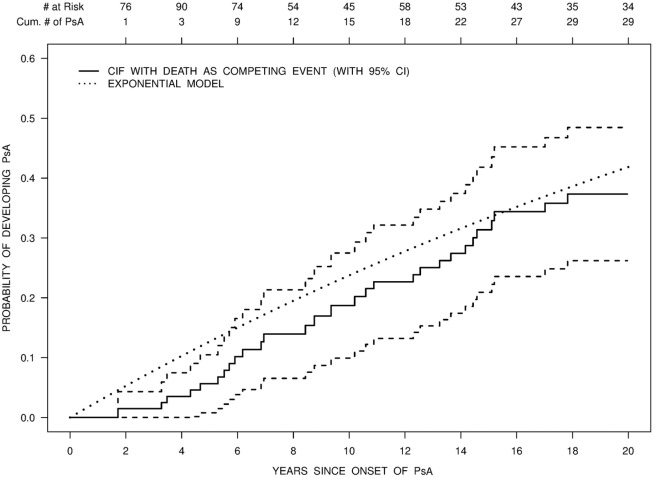
The estimated probability of developing psoriatic arthritis (PsA) over 20 years is shown as the cumulative incidence function (CIF) (with 95% confidence interval [95% CI]) for the onset of psoriasis, with death as a competing event. The estimated cumulative probability of PsA based on an exponential model is also shown. Cum. = cumulative.
Predictors of PsA in patients with psoriasis
Baseline predictors
The following baseline variables were associated with an increased risk of developing PsA in univariate analysis (each model was controlled for age at onset of PsA and stratified by sex, with use of the duration of psoriasis as a left‐truncation time): nail pitting (relative risk [RR] 2.21, P = 0.007), psoriasis severity (RR 3.86 in those with severe psoriasis and RR 1.35 in those with moderate psoriasis, Pglobal = 0.05), low level of education (RR 0.36 in those with university/college education versus high school incomplete and RR 0.54 in high school graduates versus high school incomplete, Pglobal = 0.08), and use of systemic retinoid medications (RR 2.80, P = 0.04). After multivariate regression analysis, only psoriasis severity (severe psoriasis RR 5.39, P = 0.006), low level of education (RR 0.22 [P = 0.005] in those with university/college education versus high school incomplete and RR 0.30 [P = 0.049] in high school graduates versus high school incomplete), and the use of systemic retinoid medications (RR 3.42, P = 0.02) remained independent predictors of PsA. The complete multivariate Cox regression models are presented in Table 3.
| Univariate analysis | Multivariate model | |||
|---|---|---|---|---|
| RR (95% CI) | P | RR (95% CI) | P | |
| Family history of psoriasis | 1.42 (0.82–2.45) | 0.21 | ||
| Family history of PsA or AS | 1.96 (0.57–6.71) | 0.29 | ||
| Level of education | 0.02 (global) | |||
| High school graduate vs. high school incomplete | 0.54 (0.17–1.66) | 0.28 | 0.30 (0.09–0.99) | 0.049 |
| University/college vs. high school incomplete | 0.36 (0.13–0.95) | 0.04 | 0.22 (0.08–0.62) | 0.005 |
| Any psoriatic nail lesions | 1.36 (0.76–2.45) | 0.31 | ||
| Nail pitting | 2.21 (1.24–3.92) | 0.007 | ||
| Nail onycholysis | 1.48 (0.83–2.65) | 0.18 | ||
| PASI score | 0.02 (global) | |||
| 10–20 vs. <10 | 1.35 (0.63–2.91) | 0.45 | 1.16 (0.50–2.64) | 0.73 |
| >20 vs. <10 | 3.86 (1.27–11.7) | 0.02 | 5.39 (1.64–17.7) | 0.006 |
| Alcohol consumption | ||||
| Social vs. none | 0.97 (0.51–1.82) | 0.92 | ||
| Daily vs. none | 1.02 (0.40–2.59) | 0.97 | ||
| Smoking status | ||||
| Current vs. never | 1.36 (0.68–2.73) | 0.38 | ||
| Past vs. never | 1.05 (0.56–1.99) | 0.87 | ||
| Recent infection | 1.02 (0.57–1.84) | 0.94 | ||
| Recent infection requiring antibiotics | 1.15 (0.53–2.49) | 0.72 | ||
| BMI | ||||
| Overweight vs. normal | 1.02 (0.50–2.10) | 0.95 | ||
| Obese vs. normal | 1.76 (0.89–3.47) | 0.10 | ||
| Depression | 0.92 (0.35–2.34) | 0.85 | ||
| Thyroid disease | 2.03 (0.90–4.57) | 0.09 | ||
| Diabetes | 0.53 (0.15–1.84) | 0.32 | ||
| Symptoms of diarrhea | 3.18 (0.88–11.4) | 0.08 | ||
| Use of anti‐TNF agents | 1.56 (0.19–12.6) | 0.68 | ||
| Use of retinoids | 2.80 (1.05–7.47) | 0.04 | 3.42 (1.24–9.44) | 0.02 |
| Postmenopausal state | 1.19 (0.40–3.53) | 0.75 | ||
- a Analyses were controlled for age at onset of psoriasis and stratified by sex. The variables inflammatory eye disease, use of methotrexate, and recent trauma were not included in this analysis due to paucity of positive responses. Patients with missing variables were excluded. RR = relative risk; 95% CI = 95% confidence interval; AS = ankylosing spondylitis; PASI = Psoriasis Activity and Severity Index; BMI = body mass index; anti‐TNF = anti–tumor necrosis factor.
A sensitivity analysis in which only confirmed PsA cases were considered as the study outcome showed similar results. In the sensitivity analysis, the following variables remained independent predictors of PsA in the multivariate regression models: severe psoriasis (RR 4.59, 95% CI 1.2–17.5, P = 0.025), low level of education (university/college education versus high school incomplete RR 0.20, 95% CI 0.06–0.62, P = 0.005; high school graduate versus high school incomplete RR 0.27, 95% CI 0.07–1.05, P = 0.059), and use of systemic retinoid medications (RR 3.28, 95% CI 1.06–10.2, P = 0.04).
Time‐dependent predictors
The same variables were then assessed as time‐dependent covariates in regression models (Table 4). The following variables were associated with an increased risk of developing PsA in univariate analysis (each model was controlled for age at onset of PsA and stratified by sex, with use of the duration of psoriasis as a left‐truncation time): nail pitting (RR 2.20, P = 0.005), uveitis (RR 25.3, P = 0.0001), thyroid disease (RR 2.27, P = 0.04), and psoriasis severity (severe psoriasis RR 4.02, P = 0.03). Multivariate regression analyses with time‐dependent variables showed that nail pitting (RR 2.51, 95% CI 1.37–4.49, P = 0.002) and uveitis (RR 31.5, 95% CI 5.06–195.8, P = 0.0002) were independent predictors of PsA. Obesity, a factor that was found to be predictive of the development of PsA in previous studies, showed only a borderline association with PsA in univariate analysis (RR 2.02, P = 0.06).
| Univariate analysis | Multivariate model | |||
|---|---|---|---|---|
| RR (95% CI) | P | RR (95% CI) | P | |
| Family history of psoriasis | 1.26 (0.73–2.17) | 0.41 | ||
| Family history of PsA or AS | 2.30 (0.78–6.82) | 0.13 | ||
| Level of education | ||||
| High school graduate vs. high school incomplete | 0.81 (0.25–2.66) | 0.73 | ||
| University/college vs. high school incomplete | 0.52 (0.18–1.51) | 0.23 | ||
| Any psoriatic nail lesions | 1.35 (0.78–2.34) | 0.29 | ||
| Nail pitting | 2.20 (1.26–3.83) | 0.005 | 2.51 (1.37–4.49) | 0.002 |
| Nail onycholysis | 1.33 (0.76–2.34) | 0.32 | ||
| PASI score | ||||
| 10–20 vs. <10 | 1.38 (0.61–3.11) | 0.44 | ||
| >20 vs. <10 | 4.02 (1.14–14.2) | 0.03 | ||
| Alcohol consumption | ||||
| Social vs. none | 0.87 (0.49–1.55) | 0.64 | ||
| Daily vs. none | 0.66 (0.24–1.82) | 0.42 | ||
| Smoking status | ||||
| Current vs. never | 1.10 (0.52–2.32) | 0.80 | ||
| Past vs. never | 1.30 (0.71–2.39) | 0.40 | ||
| Recent infection | 0.96 (0.52–1.77) | 0.89 | ||
| Recent infection requiring antibiotics | 0.88 (0.39–1.98) | 0.75 | ||
| Recent trauma | 1.39 (0.40–4.85) | 0.61 | ||
| BMI | ||||
| Overweight vs. normal | 1.65 (0.79–3.44) | 0.18 | ||
| Obese vs. normal | 2.02 (0.97–4.24) | 0.06 | ||
| Uveitis ever | 25.3 (4.93–130.2) | 0.0001 | 31.5 (5.06–195.8) | 0.0002 |
| Depression | 0.84 (0.37–1.89) | 0.66 | ||
| Thyroid disease | 2.27 (1.04–4.95) | 0.04 | ||
| Diabetes | 0.42 (0.13–1.43) | 0.17 | ||
| Symptoms of diarrhea | 2.95 (0.79–11) | 0.11 | ||
| Use of methotrexate | 0.60 (0.14–2.53) | 0.48 | ||
| Use of anti‐TNF agents | 1.84 (0.67–5.02) | 0.24 | ||
| Use of retinoids | 2.17 (0.88–5.32) | 0.09 | ||
| Postmenopausal state | 0.75 (0.23–2.41) | 0.63 | ||
- a Analyses were controlled for age at onset of psoriasis and stratified by sex. Patients with missing variables were excluded. RR = relative risk; 95% CI = 95% confidence interval; AS = ankylosing spondylitis; PASI = Psoriasis Activity and Severity Index; BMI = body mass index; anti‐TNF = anti–tumor necrosis factor.
A sensitivity analysis considering only confirmed cases of PsA as the study outcome showed similar results; however, the 95% CIs were wider due to a reduction in the number of events and reduced precision. Nail pitting (RR 2.39, 95% CI 1.27–4.53, P = 0.007) and uveitis (RR 55.7, 95% CI 6.99–442.9, P = 0.0001) were independent predictors of PsA in the sensitivity analysis.
DISCUSSION
In this study, we have summarized the data from 8 years of follow‐up of a prospective cohort of psoriasis patients who did not have arthritis at baseline. This unique cohort was used to investigate the incidence and markers for high risk of PsA. The estimated annual incidence was 2.7 cases of PsA per 100 psoriasis patients, an estimate that is higher than has been reported previously in the literature. Furthermore, there was no evidence to suggest that the rate of PsA development changes over time. Several risk markers for PsA were identified. These can be classified into factors related to psoriasis phenotype (nail pitting, severe psoriasis, and use of systemic retinoids), extraarticular manifestations (uveitis), and demographics (low level of education).
It is widely accepted that psoriasis and PsA are tightly linked. However, the nature and underlying mechanisms connecting these conditions remain poorly understood. Most studies that assessed the occurrence of PsA in patients with psoriasis estimated the point prevalence of PsA using cross‐sectional study designs. Based on those reports, the prevalence of PsA in psoriasis patients ranged widely, being largely dependent on the source of recruitment, with population‐based studies reporting a prevalence of PsA of ∼8% and hospital‐based studies that recruited patients from dermatology clinics reporting a prevalence of ∼30% 2, 11, 26-28. Consistent with the latter estimates, in our study, in which participants were carefully assessed for any sign of arthritis, the estimated incidence of PsA was much higher than was previously reported in population‐based studies. We found that the annual incidence of PsA was 2.7%, compared to an incidence rate of 5.1% at 20 years in a retrospective study from Rochester, Minnesota 4. This wide variation in estimates of PsA occurrence across different studies has several causes. First, methods of case ascertainment vary, and range from self‐report or screening questionnaire to rheumatologic assessment. This may explain the variation, as it has been shown that a significant proportion of psoriasis patients may have unrecognized PsA that is diagnosed only after a careful rheumatologic assessment 2, 28. Therefore, a significant proportion of patients with PsA who may possibly have milder disease escape medical attention, which may lead to underestimation of the occurrence of PsA in population‐based studies that rely mostly on administrative databases for case ascertainment. Additionally, differences in the source population may account for the higher rate of PsA in hospital‐based studies, in which participants tend to have more severe psoriasis, a factor that has been linked to a higher risk of developing PsA both in the present study and in other studies.
There is great interest in identifying risk markers for PsA in psoriasis patients. These may assist in the understanding of the underlying pathophysiologic mechanisms of PsA and facilitate the development of a prediction algorithm for PsA. In this study, we confirmed 2 of the previously suggested risk markers for PsA, psoriatic nail lesions and psoriasis severity. Nail psoriasis has been consistently associated with a higher risk of having PsA among psoriasis patients 4, 9, 10. The close anatomic link between the nail and a network of fibers in the entheses connecting to the extensor tendon and collateral ligaments, along with the enthesis organ hypothesis suggested by McGonagle and colleagues 29, may explain this association. Results of a previous study suggested a stronger association between onycholysis and PsA 30. In contrast, in our study, only the presence of nail pitting was a predictor of the development of PsA. Severe psoriasis was associated with a higher risk of having PsA in previous studies 4, 11. Our results confirmed that association.
Additionally, the use of systemic retinoids, a factor that also was predictive of the development of PsA in our study, may be considered to be a marker for more severe skin disease. Psoriasis and PsA share several proinflammatory pathways; it is possible, therefore, that more severe skin disease signals a higher systemic burden of inflammatory response as a result of shared susceptibility genes and/or environmental factors that may eventually result in the triggering of PsA. Alternatively, the larger affected body surface area may provide a wider port of entry for the skin microbiome to interact with the immune system, which may play a role in the development of PsA.
We have identified several additional novel predictors of PsA in our study. Uveitis and low level of education were associated with a higher risk of developing PsA. Uveitis is one of the characteristic extraarticular manifestations of SpA that affects ∼20% of patients 31. The occurrence of uveitis is associated mostly with axial involvement, and its frequency tends to be lower in patients with PsA compared to those with AS 31-33. In our study, the occurrence of uveitis was predictive of the development of PsA; however, it should be noted that, due to the small number of events, the 95% CIs were wide. Two of the 3 patients with uveitis who developed PsA had predominantly axial involvement.
The association between lower level of education and PsA is harder to explain. A lower level of education is a marker of lower socioeconomic status, which has been associated with lifestyle habits that may increase PsA risk. Some of the lifestyle habits, such as smoking and alcohol consumption, were measured in this study, while others were not. A lower level of education may also be associated with blue‐collar occupations, a factor that has been linked with SpA 14, 34. This association will require a more comprehensive assessment of potential underlying factors.
This study has several limitations. First, generalizability of the study may be somewhat limited, as the majority of the participants were recruited from dermatology clinics, leading to overrepresentation of patients with moderate‐to‐severe psoriasis and, possibly, patients with longer duration of psoriasis. Additionally, due to the study design, patients who developed PsA prior to or concurrent with the diagnosis of psoriasis were excluded. Therefore, our incident cases of PsA may be overrepresentative of a certain subtype of PsA, such as that associated with HLA–C*06, which has been linked with severe psoriasis and long duration between the onset of psoriasis and PsA 35.
Another limitation may be the relatively small sample size of the study, which limits our ability to draw conclusions about negative results. For example, the association between obesity and the risk of developing PsA was confirmed in 2 large cohort studies 12, 13. In the present study, the association between obesity and PsA was of borderline statistical significance. However, the effect size was similar to that observed in previous studies, suggesting that limited power may explain the negative results.
In summary, in this prospective cohort study, the incidence of and risk factors for PsA in patients with psoriasis were assessed. Overall, it is likely that the true incidence of PsA in patients with psoriasis, particularly those attending dermatology clinics, is higher than previously reported. This highlights the role of dermatologists as key players in identifying psoriasis patients who are at higher risk of developing PsA. Recognition of several clinical markers may assist in the identification of patients at risk of developing PsA.
AUTHOR CONTRIBUTIONS
All authors were involved in drafting the article or revising it critically for important intellectual content, and all authors approved the final version to be published. Dr. Gladman had full access to all of the data in the study and takes responsibility for the integrity of the data and the accuracy of the data analysis.
Study conception and design
Eder, Rosen, Lee, Chandran, Cook, Gladman.
Acquisition of data
Eder, Haddad, Rosen, Chandran, Gladman.
Analysis and interpretation of data
Eder, Haddad, Rosen, Lee, Chandran, Cook, Gladman.